Radiopacity: I can see clearly now
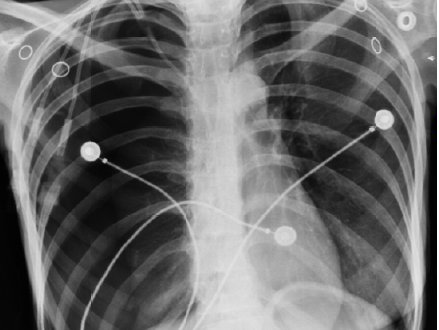
Radiopacity (or radiodensity) is the ability of a material or device to block or obstruct the passage of electromagnetic photons, normally in the form of X-rays. On an photographic X-ray image, a material with more radiopacity than the background will appear brighter than the background due to the unexposed emulsion not developing on the image. For historical reasons this relationship is preserved for modern digital images as well. In general, the more dense a material is, the higher its radiopacity, although the nature of the specific atoms present (how electron dense they are) also plays a role. As such, metals and ceramics tend to have higher radiopacity than plastics and fluids. Lead, which has a density of 11.8 g/ml, is one of the more dense metals, and is why it is used as a shielding material for X-rays. The opposite of radiopacity is radiolucency.
Device manufacturers will often incorporate metals such as tantalum, tungsten, and stainless steel into devices for temporary or permanent implantation. Salts such as barium sulfate, zirconium oxide, and bismuth are also used to render plastics radiopaque. Increasingly, regulatory agencies and device manufacturers are requiring quantification of the degree of radiopacity in medical devices to assure that these devices exhibit sufficient radiodensity for their application.