Do I have to do chemical characterization?
Overheard at a recent conference: “Oh, our device isn’t a permanent implant, so we don’t need to do extractables testing.”
Is that right? If a device is implanted for less than 30 days (or even less than 24 hours), is extractables and chemical characterization unnecessary?
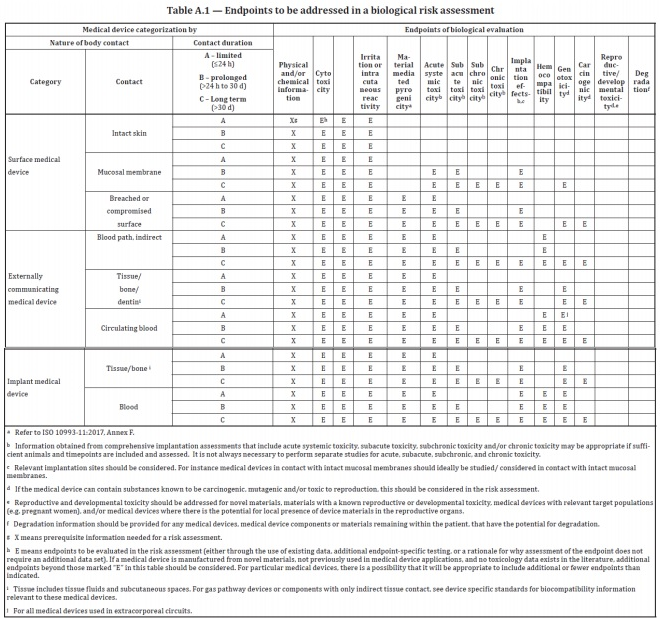
For clarity on this issue, we turn to ISO 10993-1:2018 and the FDA 2016 guidance on biocompatibility assessment. Each of these references contains a table which breaks down the biocompatibility evaluation endpoints associated with different implantation durations and the nature of body contact. Even in the case of a “limited” contact duration of less than 24 hours, these documents indicate that a biocompatibility assessment be performed. What does that entail, however?
The first step in performing any extent of biocompatibility assessment is to compile a detailed understanding of the physical/chemical properties and chemical composition of the device. Only once this information is in hand can an evaluation and risk assessment be performed with regard to toxicological endpoints such as carcinogenicity, genotoxicity, etc. For short term implants, the number of biocompatibility evaluation endpoints is generally less extensive than long term implants.
If the chemical composition of the device (both the raw materials as well as any manufacturing residues) are unknown or have not been previously characterized, an extractables assessment is typically necessary to determine the composition. Unless the device manufacturer has previous experience characterizing the material/manufacturing process by extractables, sufficient information on the device composition (including impurities, manufacturing residues, etc) is generally not available for this to be a pure paper exercise.
Note that from an analytical perspective, given the short term nature of the device, the analytical evaluation threshold (AET) employed will be less stringent than for a permanent implant. This means that when evaluating extractables data from techniques like GC-MS, and LC-MS, the number of peaks which must be identified and submitted for toxicological risk assessment is significantly lower than for a permanent implant–translating to generally lower cost and faster turnarounds.