Analysis of Vitamin E residues
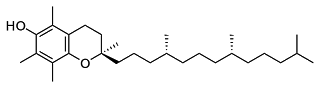
The increasing prevalence of Vitamin E, a naturally-occuring antioxidant, in medical grade plastics has resulted in a need for analysis methods that can track the effect of processing on this compound. Vitamin E is effective as an antioxidant due to the hydroxyl group sitting on the aromatic ring at one end of the molecule. This hydroxyl group can readily lose a hydrogen and capture a free radical, a culprit in oxidation reactions. The free radical is thereafter stabilized by the Vitamin E molecule.
When processing medical grade plastics, the plastics are exposed to high heats and pressures during molding. Additionally, cleaning agents and ionizing radiation are often used to clean and sterilize the finished components. All these steps can potentially modify some of the Vitamin E molecules.
As a result, medical device manufacturers are usually required to identify and quantify these modified molecules to ensure both that they are safe for in vivo use, and that the material remains adequately oxidatively stabilized. Researchers at Cambridge Polymer Group have developed a series of assays to analyze potential transformation products of Vitamin E, using a combination of chromatography and spectroscopy. Additionally, we have a series of analyses to test the oxidation resistance of stabilized plastics. All these methods have been successfully used for regulatory submissions.