Recent News
SPE Mini Tech 2024, February 5-6

On Tuesday, February 6, Cambridge Polymer Group scientists presented two talks during the 1:30-3 p.m. Characterization & Validation of Medical Devices session:
- Particulate Testing for Additive Manufactured Medical Devices, Dr. Becky Bader
- Lab X Found WHAT In My Extracts? Best Practices in Unknowns Identification in Extractables Testing, Dr. Michael Young
PharmaEd Extractables & Leachables West - November 2-3

Dr. Rebecca Bader, CPG's Associate Director of Chromatography and Biocompatibility Specialist, presented "Lab X Said What Is in My Medical Device!? Accurate Identification of Unknown Compounds from Non-Targeted Screening."
MDM Minneapolis, Oct 10-11

September 20-21 - BIOMEDevice Boston 2023
BIOMEDevice in Boston, Booth #1120.
September 19 - Massachusetts Manufacturing Mashup
Worcester Polar Park at the FORGE tabletop exhibition space.
June 13 -15 - MD&M East 2023

May 9 - ASTM F04 Workshop on Setting Acceptance Limits for Cleaning Validation of Medical Devices
MDM West - February 7-9

Cleaning Validation Summit 2022 - December 1-2

CPG President Stephen Spiegelberg serves as a panelist at PharmaEd's Cleaning Validation Summit 2022, December 1-2 in San Diego, California.
BIOMEDevice Boston - September 28-29

Stop by Booth 1120 at BIOMEDevice, Boston Convention & Exhibition Center, 415 Summer St., Boston.
Massachusetts Manufacturing Mash-Up - September 16
Join us at the 2nd Massachusetts Manufacturing Mash-Up!
FORGE - Material Selection for Physical Product Development - May 17

CPG President Stephen Spiegelberg participated on the FORGE panel about choosing materials wisely for increased product success in medical or harsh environments. UMass Lowell, 110 Canal Street, 3rd flr, Lowell. 5-6:30 p.m.
ARVO - May 1-4

VP of Research Gavin Braithwaite presents a poster on "
VP of Research Gavin Braithwaite presented a paper at ARVO 2022 on "A Temporary Hydrogel Vitreous Substitute in Support of Retinal Detachment Repairs" in Denver.
April 12-14 - MDM West 2022

March 28-30 - SOT 2022
December 7-9 - MDM East 2021

Visit us at MDM East 2021, Booth 1630 at the Javits Center in NYC.
December 1 - 2021 MRS Fall Meeting

In Session SB12.05, CPG research scientist Mimoza Xheka presents "Biodegradable poly(vinyl alcohol) Hydrogels as a Temporary Retinal Tamponade" at the Hynes Convention Center, Boston, from 11:45 a.m.-12 p.m.
September 28 - Massachusetts Manufacturing Mashup 2021

Cambridge Polymer Group exhibited at the Massachusetts Manufacturing Mash-Up, Polar Park, Worcester, MA on September 28.
September 21-22 - BIOMEDevice 2021

CPG exhibited at BIOMEDevice 2021, Booth 439.
August 10 -12 - MDM West 2021
August 4 - 5 - CPG MOVES TO Woburn

The 117% increase in lab space will allow us to meet the growing demand for our analytical testing and research & development services.
April 28 - MIT POLYMER DAY 2021
Due to the ongoing COVID-19 pandemic, Polymer Day 2021 will be taking place in a completely virtual format.
February 11-13 - MD&M West 2020, Booth 435

November 3-6, 2019 - AAPS PharmSci 360, Booth 1933

October 28-29, 2019 - Generis American Medical Device Summit, Booth 51

CPG VP Gavin Braithwaite presented A New Vision for Biomaterials: Crafting Biodegradable Hydrogels for Ophthalmic Devices:
- Discussing multi-discipline requirements for the development of soft-solid systems in biomedical applications
- Reviewing design processes for soft-solids from product requirements to final device
- How to track soft-solids in vivo
October 9-10 - PharmaEd's Cleaning Validation Summit 2019

CPG President Stephen Spiegelberg presented ASTM Standards in Medical Device Cleaning Validation at the PharmaEd Cleaning Validation Summit 2019.
September 17 - Greentown Learn Manufacturing Initiative Supplier & Innovation Showcase

Cambridge Polymer Group sponsored Greentown Learn Manufacturing Initiative Supplier & Innovation Showcase.
June 11-13 - MDM East 2019, Booth 646

May 15-16 - BIOMEDevice 2019, Booth 115

May 14 - ASTM WORKSHOP ON ACCELERATED AGING METHODS AND TESTING TECHNIQUES FOR MEDICAL DEVICES

CPG President Stephen Spiegelberg co-chaired an accelerated aging workshop, sponsored by ASTM Committee F04 on Medical and Surgical Materials and Devices, in Denver, Colorado.
CPG Research Scientist Norma Turner's presented "Differentiating the effects of temperature and elevated oxygen pressure on accelerated aging of UHMWPE."
February 5-7 - MD&M West 2019, Booth 530

November 4-7 - AAPS PharmSci 360 2018, Booth 1343

AAPS PharmSci360, Walter Washington Convention Center, DC
October 24-25 - AMI's Polymer Testing & Analysis 2018

Cambridge Polymer Group attended the 2018 American Medical Device Summit on October 24-25 in Lombard.
September 12-13 - PharmaEd's Cleaning Validation Summit 2018
CPG talks at PharmaEd's summit in Philadelphia:
Wednesday, September 12
CPG President Stephen Spiegelberg presented "ASTM Standards in Medical Device Cleaning Validation" at 10:40 a.m.
Thursday, September 13
CPG Research Scientist Adam Kozak presented "Development of Chromatography Assays For Cleaning Studies of Medical Devices" at 8:45 a.m.
September 11-12 - AMI's Polymer Testing & Analysis 2018
CPG Research Scientist Adam Kozak presented "Size (and Shape) Matter: Challenges and Strategies for Difficult Molecular Weight and Polymer Architecture Analysis" on September 11, Pittsburgh Marriott City Center, 112 Washington Pl, Pittsburgh, PA.
June 12-14 - MDM East 2018

May 7-11 - NPE 2018

Stop by Booth S30200 in the South Hall Level 1 - Expo Hall at NPE 2018: The Plastics Show, Orange County Convention Center, 9800 International Dr, Orlando. CPG Research Scientist Adam Kozak presents "Degradation Products of Medical Devices in Complex Biological Environments" on Wednesday, May 9, from 3:30-4 p.m. at the ANTEC Technical Program @NPE 2018.
April 18-19 - BIOMEDevice 2018

April 11 - MIT Polymer Day
Cambridge Polymer Group proudly sponsored Polymer Day 2018 at MIT.
February 6-8, 2018 - MDM WEST

November 28-30, 2017 - MATERIALS RESEARCH SOCIETY FALL MEETING & EXHIBIT

October 12, 2017 - Extractables & Leachables Symposium for Drugs & Devices
CPG researcher Adam Kozak spoke at Eurofins' Extractables & Leachables Symposium for Drugs & Devices in Lancaster, PA.
October 2-3, 2017 - Cleaning Validation Summit
CPG President Stephen Spiegelberg spoke at PharmaEdResources' Cleaning Validation Summit 2017 in Philadelphia on Tuesday, October 3.
September 27-28, 2017 - American Pharma Outsourcing Summit 2017

CPG exhibited at the American Pharma Outsourcing Summit 2017, Doubletree by Hilton Boston North Shore, Danvers, Massachusetts.
September 19-20, 2017 - AMI's Polymer Testing & Analysis
CPG exhibited at Polymer Testing & Analysis at the Pittsburgh Marriott City Center.
September 14, 2017 - Contract Pharma

CPG exhibited at Contract Pharma.
August 14, 2017 - Cambridge Polymer Group Awarded $225,000 SBIR Grant from the National Institutes of Health
Cambridge Polymer Group’s team of hydrogel scientists are developing a hydrogel that can be injected as a liquid through a fine needle or cannula, and then gels in the eye. This hydrogel will assist in keeping the retina attached to the back wall of the eye, and will then degrade over time, obviating the need for a second surgery. The hydrogel should also allow vision through the eye while it is in place, resulting in greater patient mobility and compliance.
June 13-15 - MDM East 2017

CPG President Stephen Spiegelberg presented a workshop on Developing a Testing Plan for Medical Device Design Verification.
May 31, 2017 - Minimizing Risk in Medical Device Material Selection

Dr. Brian Ralston discussed material selection considerations and process, selection of performance specifications, material screening, full assembly or sub-assembly prototype on candidate materials, and simulated testing of device. To view the webinar recording, click here.
May 10 - UNH Bioengineering Symposium 2017

Dr. Gavin Braithwaite was an invited speaker and judge at the annual UNH Bioengineering Symposium. He presented "Designing Medical Devices Using Soft Materials."
May 8-10 - ANTEC/SPE 2017

CPG's Dr. Brian Ralston presented "Medical Plastics: Where to Start with Material Selection" at ANTEC 2017.
May 3-4 - BIOMEDevice 2017
Cambridge Polymer Group exhibited at BIOMEDevice, Boston Convention & Exposition Center.
March 2017 - CPG Receives ISO 17025:2005 Accreditation
Cambridge Polymer Group is pleased to announce that the American Association for Laboratory Accreditation (A2LA) has approved our company for ISO 17025:2005 accreditation.
February 23, 2017 - Cleanliness in Medical Devices Webinar

CPG's webinar on cleanliness in medical devices included examples of what happens when cleaning processes are not properly verified and validated, how to establish the number of samples to test, how to test for device cleanliness, and how to establish acceptable residue limits.
Dr. Stephen Spiegelberg is the chairman of the ASTM task group on Medical Device Cleanliness, and Dr. Gavin Braithwaite regularly consults on cleaning issues in the medical device area.
To listen to a recorded version of the webinar, register here.
March 20 - 22, 2017 - CPG Exhibits at ORS, San Diego

January 2017 - CPG Receives Patent for Patient Positioning
CPG researchers, along with orthopedic surgeons from the Massachusetts General Hospital, have received a U.S. patent for a low cost navigation system designed to assist surgeons in aligning the acetabular cup used in hip replacement surgeries. The patent was issued as patent number 9,554,731.
January 24 -25, 2017 - CPG President Speaks at International Standards Meeting about Cleanliness in Medical Devices

Scientists from the FDA, American Dental Association, medical device companies, and contract research laboratories were asked to present to Peruvian medical device regulators and manufacturers on how ASTM standards benefit their organizations in helping to get safe and effective medical devices into the patients who need them. Peru has a keen interest in improving their adoption of standards for material and product testing in order to market safe products both in Peru and outside of Peru.
Dr. Stephen Spiegelberg from Cambridge Polymer Group spoke about his ASTM task group on Medical Device Cleanliness, and provided examples of what can go right and what can go wrong when ASTM standards are not properly used, particularly with respect to validation of medical device cleaning lines.
Read more in this ANSI article and this ADA article.
November 29 - December 1, 2016 - MRS Fall Meeting & Exhibit

Cambridge Polymer Group exhibited at the 2016 Material Research Society Fall Meeting & Exhibit.
November 1, 2016 - CPG ACHIEVES ISO 9001:2015 CERTIFICATION

In November 2016, CPG successfully transitioned from ISO 9001:2008 to the updated ISO 9001:2015 standard. Read more about CPG's new ISO certification.
October 28, 2016 - FDA Workshop on Refurbishing of Medical Devices Performed by Third-Party Entities and Original Equipment Manufacturers
CPG President Stephen Spiegelberg spoke at an FDA workshop on "Refurbishing, Reconditioning, Rebuilding, Remarketing, Remanufacturing, and Servicing of Medical Devices Performed by Third-Party Entities and Original Equipment Manufacturers" in Silver Springs, Maryland.
June 14-16, 2016 - MD&M East

Cambridge Polymer Group exhibited at MD&M East, the Javits Convention Center, NYC, Booth 175. For more info, click here.
May 17-22, 2016 - World Biomaterials Congress

CPG researcher Adam Kozak presented a poster on a cardiovascular device designed to reduce mitral valve regurgitation following a ventricular infarction. For more info on the World Biomaterials Congress in Montreal, click here.
April 13-14, 2016 - CPG Exhibits at BIOMEDevice 2016
March 30, 2016 - CPG Sponsors MIT Polymer Day

Cambridge Polymer Group sponsored MIT's 6th Annual Polymer Day on March 30th, 2016. CPG scientists Gavin Braithwaite and Brian Ralston were judges at the poster contest, where MIT researchers presented their latest research. For more on MIT's Polymer Day, click here.
March 22, 2016 - Patent on Mosaicplasty Implants Issued to CPG Researchers
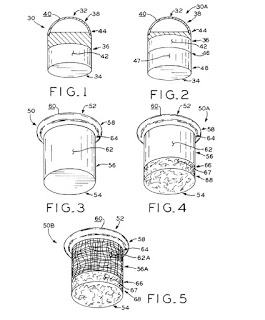
US Patent 9,289,302 was issued on March 22, 2016 to CPG researchers. The patent describes designs and methods for making implants for focal cartilage tears in load bearing implants, such as hips and knees. The construct designs make use of a surgical procedure termed mosaicplasty, whereby a plug of healthy osteochondral plug is removed from a non-load bearing portion of the joint, and placed in a drilled out cavity in the focal defect. In this patent, synthetic plugs are created based on hybrid structures of polymers and metal repair the focal defects.
March 5-8, 2016 - CPG Exhibits at ORS 2016 Orlando

March 2016 - Notch Fatigue in UHMWPE

CPG researcher Adam Kozak co-authored a Journal of Mechanical Behavior of Biomedical Materials article. Along with co-authors from UC-Berkeley (Ansari, Gludovatz, Ritchie, and Pruitt), these researchers investigated the sensitivity of ultra high molecular weight polyethylene (UHMWPE) to fatigue when stress concentration sites are present in the form of notches. The authors investigated 3 formulations of UHMWPE commonly used in hip or knee implants today, and found that the sensitivity to crack propagation resulting from fatigue was more sensitive to microstructure, such as crosslink density, rather than specifics of the notch geometry.
January 2016 - CPG Acquires Herzan AVI-400 for SEM

Over two years ago, CPG purchased a Zeiss EVO LS15 environmental scanning electron microscope. This instrument has become a work-horse for root-cause analysis, surface topographies and (through the attached Oxford EDS) elemental mapping. With the large chamber and capability to run in “wet mode” (hydrated at elevated pressures) it has also proved invaluable at imaging hydrogel structures and friable materials such as foods and tissues.
As part of an effort to improve the resolution of the instrument, CPG recently procured a Herzan AVI-400 antivibration solution. The entire SEM system sits on this active vibration damping system, removing virtually all of the building and environmental vibration. We are delighted that the Zeiss now achieves resolutions close to the ideal specifications of the instrument.
January 2016 - The Rheology of Hyaluronic Acid Used to Treat Osteoarthritis

Recently researchers at Cambridge Polymer Group in collaboration with OrthogenRx coauthored a paper on biosimilar medical devices and methods for supporting performance similarities in specifically viscosupplementation. In this paper, we compare an existing material cleared for sale in the US, against an ex-US device intended to replicate the performance of the existing material as a “generic device” equivalent to generic drugs. The ability to market generic devices has only recently been made possible through changes in the regulatory mechanisms in the US, and this paper discusses this context and describes analytical methods for comparing the performance of viscosupplementation materials. Read the paper here.
December 1-3, 2015 - CPG Exhibits at MRS 2015 Boston

October 27, 2015 -3rd Edition of UHMWPE Biomaterials Handbook

This edition contains the history of ultra high molecular weight polyethylene and its use in hip and knee arthroplasty. The new edition contains multiple chapters addressing analytical testing techniques to characterize UHMWPE, wear testing, accelerated aging, antioxidant effects, and advances in UHMWPE processing and formulation development.
CPG researchers Braithwaite, Kozak, and Spiegelberg contributed a chapter on characterization techniques on UHMWPE, including details on fatigue crack propagation testing, true stress-true strain measurements, and electron spin resonance spectroscopy.
Link to purchase 3rd edition of the UHMWPE Handbook
October 22, 2015 - Fatigue Crack Propagation Testing in UHMWPE
This poster presentation discusses the comparison of fatigue crack propagation testing per ASTM E647 using a new optical system developed at Cambridge Polymer Group to monitor crack growth in an automated manner. The presentation shows results from two laboratories using the system. Presented at the 7th semi-annual UHMWPE Conference on October 22, 2015 in Philadelphia, PA.
September 15-16, 2015 - Medical Grade Polymers 2015

Dr. Yuri Svirkin, CPG Research Scientist, and Mr. Adam Kozak, CPG Research Scientist, presented "Novel Material Development for Medical Devices."
March 11, 2015 - MIT Celebrates Polymer Day

On March 11, MIT's Program in Polymers and Soft Matter hosted their 5th Annual Polymer Day. A poster session was held in the morning, and two CPG scientists, Dr. Yuri Svirkin and Adam Kozak, acted as judges and awarded prizes to the top posters.

Cambridge Polymer Group received an appreciation award for three years as a Polymer Day Sponsor. Dr.Yuri Svirkin (left) and Research Scientist Adam Kozak (center) accepted the award.
December 2-4, 2014 - CPG Exhibits at 2014 MRS Fall Meeting & Exhibit

October 16, 2014 - MD&M Chicago

CPG researcher Stephen Spiegelberg presented "Failure Analysis of Medical Plastics."
August 2014 - CPG Acquires Variable Pressure SEM

CPG recently acquired a variable pressure scanning electron microscope (SEM). This system is useful for failure analysis of components, in that the imaging is non-destructive. Additionally, the CPG system has a cold stage, which allows the control of relative humidity and temperature in the chamber, which is useful for imaging water-containing samples such as tissue and hydrogels. The system acquired also has a large capacity chamber enabling in most cases visualization on entire devices and components. More info
June 10-12, 2014 - CPG Exhibits at Medical Device and Manucturing East

June 2014 - CPG Acquires an ESR for Analysis of Free Radicals
Electron Spin Resonance is used to determine concentrations of free radicals in foods and materials. In the beer industry it is invaluable as a measure of how prone the beer is to "staling" where beer-derived free-radicals ultimately result in the production of carbonyl end products that result in the distinctive "off" flavor of old beer. In the orthopaedic industry free radicals are induced by the crosslinking, or other manufacturing process, and therefore tracking these radicals is critical for determining ultimate stability of the materials. CPG's acquisition of an ESR, along with the extensive calibration and validation we have performed, provides a valuable tool in determining likely stability of materials.
May 20, 2014 - US Patent Issued to CPG
 CPG was issued US Patent 8728379 in May 2014. This patent describes methods of making wear resistance, oxidatively stable polyethylene for orthopedic implants. The technology involves irradiating ultra high molecular weight polyethylene (UHMWPE) which contains Vitamin E as an antioxidant. This patent is available for license. For more information, please visit our web site.
CPG was issued US Patent 8728379 in May 2014. This patent describes methods of making wear resistance, oxidatively stable polyethylene for orthopedic implants. The technology involves irradiating ultra high molecular weight polyethylene (UHMWPE) which contains Vitamin E as an antioxidant. This patent is available for license. For more information, please visit our web site.
April 28, 2014 - CPG Publishes Journal Article on Vitamin E Measurement Technique for UHMWPE
 CPG researchers recently published a paper on a sensitive technique to quantify Vitamin E in UHMWPE used for orthopedic applications. The article is published in the Journal of Biomedical Materials Research.
CPG researchers recently published a paper on a sensitive technique to quantify Vitamin E in UHMWPE used for orthopedic applications. The article is published in the Journal of Biomedical Materials Research.
January 28, 2014 - CPG Receives Patent for Highly Lubricious and Tough Hydrogels
CPG inventors were assigned US patent 8,637,063 for methods of making highly lubricious hydrogels that may be suitable for joint replacement or bearing surfaces.

These hydrogels are manufactured in a novel manner by first forming an organogel, and then converting this organogel to a hydrogel. This technology is available for license, learn more here.
January 2014 - CPG Acquires a TGA with FTIR Analysis Accessory
CPG recently acquired a thermal gravimetric analyzer. This tool provides the ability to separate compounds from a material by their evaporation point, and also examination activation energies and ash content. In conjunction with the FTIR accessory, we can also determine the molecules begin involved. This combination of tools expands our ability to perform deformulation work, particularly in complex polymeric systems. Using the TGA in a "pyrolysis" mode allows identification of individual polymeric species, as well as additives in the final composition.
2013 Fall MRS Research Presentation

CPG researchers Yuri Svirkin, Adam Kozak, and Gavin Braithwaite presented their work on thiol-modified PVA hydrogels at the Fall Materials Research Society Meeting in Boston on December 3rd (Paper E5.09). Learn more about 2013 MRS Fall Meeting here.
October 29, 2013 -CPG Receives Patent for Gradient Antioxidant UHMWPE

CPG inventors, along with inventors from the Massachusetts General Hospital, were assigned US Patent 8,569,395 for methods of making oxidation-resistant crosslinked polymeric materials. This patent discusses methods of incorporating antioxidants into UHMWPE to allow spatial control of crosslinking and antioxidant distribution, and has applications for total hip, knee, and shoulder arthroplasties. This technology is available for license. Learn more here.
October 15-16, 2013 (Houston, TX). MedTech Polymers Conference
Dr. Stephen Spiegelberg presented a paper on failure analysis of medical devices, including testing methods. More Info
October 2013 - CPG aquires new Impact Tester

The recently purchased CEAST 9050 (Instron) impact tester can perform Izod testing in compliance with both ASTM D256 and UHMWPE testing per ASTM F648.
September 2013 - CPG Scientist Elected to post in ASTM committee

CPG researcher Dr. Stephen Spiegelberg was recently elected to the post of Recording Secretary for ASTM Committee F04 “Medical and Surgical Materials and Devices”. Click here for more information on the committee.
August 2013 - Cambridge Polymer Group announces the opening of a new West Coast office
 Cambridge Polymer Group has expanded its operation with the opening of a West Coast office to support growing demand for materials consultation. Office manager: Ayyana Chakravartula, PhD. (617) 629-4400 Ext. 23.
Cambridge Polymer Group has expanded its operation with the opening of a West Coast office to support growing demand for materials consultation. Office manager: Ayyana Chakravartula, PhD. (617) 629-4400 Ext. 23.
August 22, 2013 - US Patent for High Crosslinked UHMWPE Issues
US Patent 8530057 "Methods for making oxidation resistance polymeric material" issued this year. This patent discusses methods of making highly crosslinked ultra high molecular weight polyethylene (UHMWPE) resistant to oxidation by incorporating vitamin E, a naturally occuring antioxidant. This material is available for licensing, and has been cleared by the FDA and notified bodies in the EU for use in hip and knee devices.
June 26-28, 2013 - Polymers and Plastics in Medical Devices Conference (San Francisco, CA)

CPG researcher Gavin Braithwaite presented a paper on hydrogel use in medical devices. The paper discusses the design of cartilage replacement technologies using hydrogels.
May 30, 2013 - Australian Patent for Highly Crosslinked UHMWPE Issues
Australian patent 2010206016 "Methods for making oxidation resistance polymeric material" issued this year. This patent discusses methods of making highly crosslinked ultra high molecular weight polyethylene (UHMWPE) resistant to oxidation by incorporating vitamin E, a naturally occuring antioxidant. This material is available for licensing, and has been cleared by the FDA and notified bodies in the EU for use in hip and knee devices.
March 8, 2013 - Program in Polymer Science and Technology - Polymer Day
MIT held its annual MIT Polymer Day in March. Cambridge Polymer Group was a sponsor, and also supplied two judges for the poster contest, where students and post-doctoral research fellows presented their work on polymer technology. More information.




